

http://www.myparkingsign.com/Parking-Space-Sign/Letter-P-Sign/SKU-K-6295.aspx http://www.narvajoesuu.ee/failid/Image/Hotel/parking_sign.png
What is Phosphorus?
http://education.jlab.org/itselemental/ele015.html
http://www.rsc.org/periodic-table/element/15/phosphorus
https://www.google.co.uk/#hl=en&q=allotrope&tbs=dfn:1&tbo=u&sa=X&ei=hdCBUP_yG8viyAHqkYHIDA&sqi=2&ved=0CBwQkQ4&bav=on.2,or.r_gc.r_pw.r_qf.&fp=9cc6ce1e3edfb4fe&bpcl=35466521&biw=571&bih=646
Phosphorus is a nonmetal element, with a symbol P and atomic number 15. Phosphorus was discovered in 1669 by Hennig Brandt, by boiling, filtering and processing buckets of urine. Now a days we obtain phosphorus from phosphate rock. It is a solid at room temperature and has three main allotropes ("Each of two or more different physical forms in which an element can exist.")
White phosphorus is poisonous and can ignite involuntarily when in contact with air (approx. 300C). It gives off smoke and glows in the dark when in contact with oxygen, this is where phosphorus got its name, which in greek means "light bearing", 'phosphoros'.
Red phosphorus is formed by heating white phosphorus to 2500C. It is not poisonous and is more stable.
Black phosphorus is the least reactive form of phosphorus
White Red (violet) Black

http://upload.wikimedia.org/wikipedia/commons/8/88/PhosphComby.jpg
What is atomic number?
http://www.ndt-ed.org/EducationResources/HighSchool/Radiography/atomicmassnumber.htm / notes
Atomic number is the number of protons in the nucleus of an atom. For example, Phosphorus has an atomic number of 15. Meaning, it is the only element that has 15 protons and an atomic number of 15.
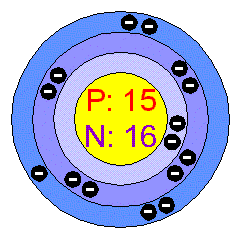 < This image shows Phosphorus's 15 negative electrons orbiting the atom's nucleus of 15 protons
< This image shows Phosphorus's 15 negative electrons orbiting the atom's nucleus of 15 protons
What is atomic mass?
http://www.windows2universe.org/physical_science/physics/atom_particle/atomic_mass.html
Atomic mass is the measurement of the mass of an atom. About 99% of an atom's mass comes from its nucleus. The atom's nucleus is made up of protons and neutrons. When you add up the number of protons and neutrons of an atom, you get the atomic mass. Phosphorus has an atomic mass of 30.973762, so approximately 31.
What contains Phosphorus?
http://www.lenntech.com/periodic/elements/p.htm
http://www.phosphatesfacts.org/faqs.asp
Fertilizers (http://www.wisegeek.com/what-is-phosphorus-fertilizer.htm): Phosphorus is a natural nutrient need for plant growth and health. It plays a role in photosynthesis, cell division and the plant's ability to use sugars and starches. Plants absorb the phosphorus through their roots. Phosphorus is found in fertilizers because it is an efficient way of supplying plants with need phosphorus.

http://1.bp.blogspot.com/_GYu5eN8tEBI/TPgOKTIa2nI/AAAAAAAAAPU/qpkZD1qhpGA/s1600/fertilizer.jpg
None-Safety Matches:
Red phosphorus is found at the "head" of matches. Phosphorus is the active ingredient that when stricken on a rough surface, ignites by frictional heat.


http://www.photo-dictionary.com/photofiles/list/833/1261matches.jpg http://en.wikipedia.org/wiki/File:Zapalky_makrofoto.jpg
Humans ( http://www.umm.edu/altmed/articles/phosphorus-000319.htm): Phosphorus is the second most abundant mineral in the human body. Calcium, being the most abundant mineral in the body, works closely with phosphorus to build strong bones and teeth.
Phosphorus helps: filter out waste in the kidneys
the body store and use energy
the growth, maintenance, and repair of all tissues and cells
produce DNA

http://futurity.org/wp-content/uploads/2010/02/teeth2.jpg
Military Applications:(http://www.fas.org/programs/bio/factsheets/whitephosphorusfactsheet.html)(http://www.globalsecurity.org/military/systems/munitions/wp.htm) Phosphorus is used in military grenades, and mortar and artillery shells. It is used to mark targets and provide a smokescreen. The phosphorus is highly active and ignites once in contact with oxygen. It bursts into flames and creates a dense white smoke.

http://static.guim.co.uk/sys-images/Guardian/Pix/pictures/2009/1/21/1232561961477/White-Phosphorus-001.jpg
http://www.rsc.org/periodic-table/element/15/phosphorus
Comments (0)
You don't have permission to comment on this page.